Testing Facility
What are the BioMeridian devices and Meridian Stress Management MSA?
How the BioMeridian Devices Work
BioMeridian Energetic Balancing - Energetic balancing with the MSAS-Professional system is done using the BioEnergetic Evaluation.
Meridian Stress Assessment - MSA - This system produces an electromagnetic frequency relating to an item being tested which is passed into the patients body via a focus antenna placed adjacent the skin, and measures the flow of micro electrical current at specific acupressure or meridian points (no needles or skin puncturing is used), which directs the practitioner to probable areas of imbalance. The Biomeridian technology is used in various health care practices around the world. It gives practitioners more information to consider, This system has been shown in a double blind study to be as good as the traditional "Elimination Diet" in identifying food intolerances which is an accurate albeit long drawn out process of identifying potential food reactions through trial and error over a 6-12 month period, Results are instant and ready for printing by the end of the test.
MSAS Professional Highly Flexible MSA platform. Can be used for quick "First Assessments" or for in-depth testing. As your practice moves forward you can add devices like the Focus and the new Epic Probe
According to European medical research, acupuncture points are related to the body's organs and organ systems. Major groups of points are connected through channels, or meridians. Twenty of these meridians begin or end on the hands and feet.
As a patient moves toward or away from health, the condition of any particular organ or system can be sensed along the meridians at representative points. As a result, stress associated with the corresponding organs can be surveyed using the indicated points.
After the initial measurements with the BioMeridian have been taken and recorded, the results can be reviewed. If stress values are above or below equilibrium, the System's extensive computer database will allow consideration of a wide range of possibilities that might help the patient regain a healthy balance. The MSAS allows consideration of thousands of herbal, homeopathic, and nutritional products.
Overall, an BioMeridian MSAS provides a completely non-invasive method for gaining valuable information about the body's vital functions. The primary objective of this procedure is to disclose patterns of stress and to provide feedback for use in a program to help restore each system and meridian to an appropriate balance.
BioMeridian MSAS-Professional does not provide a medical diagnosis. If patients suspect that they need medical intervention, they should consult their physicians who can provide medical diagnosis and prescribe appropriate treatment regimens.
For the Full Double Blind Studies Utilising the MSAS Pro Please Click Here
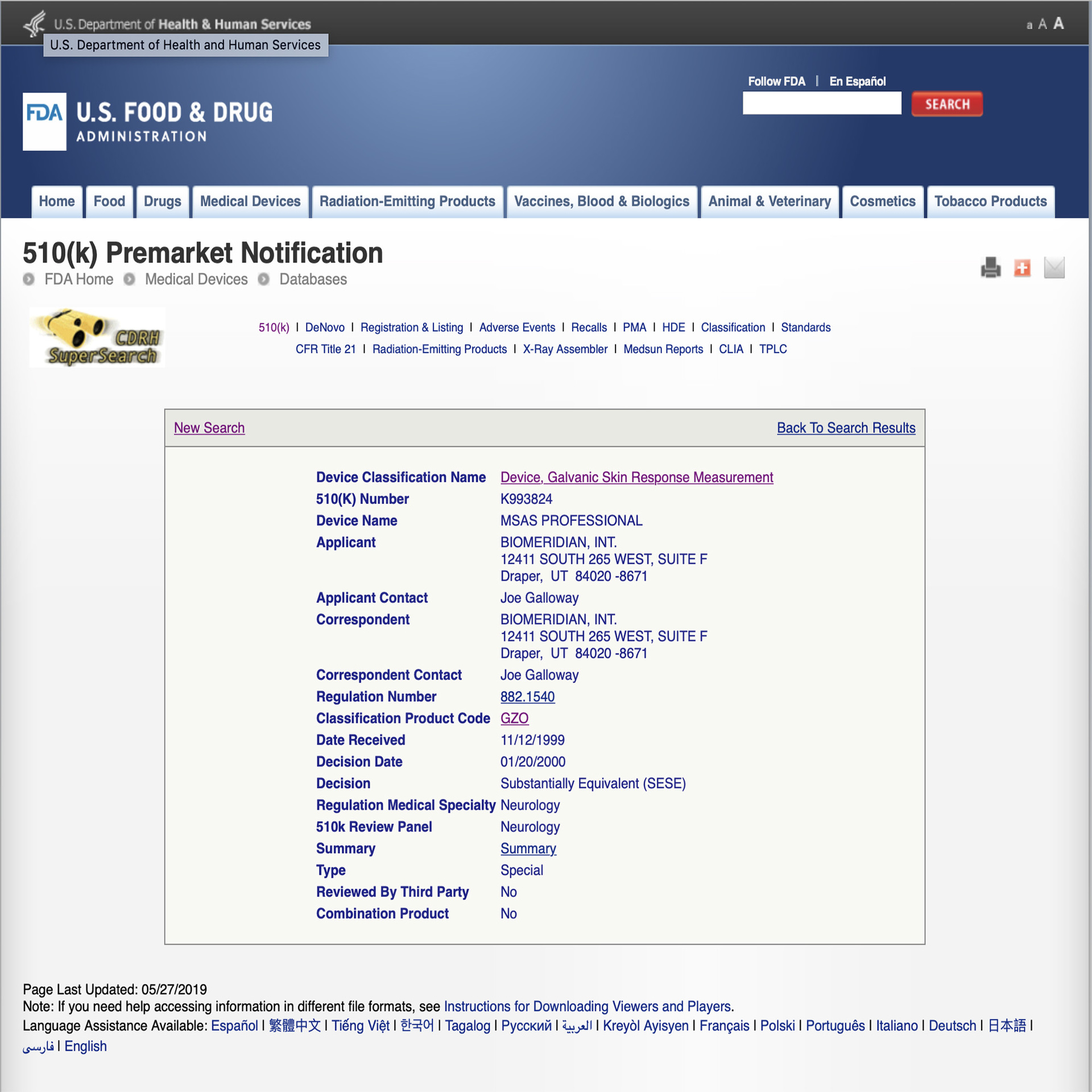
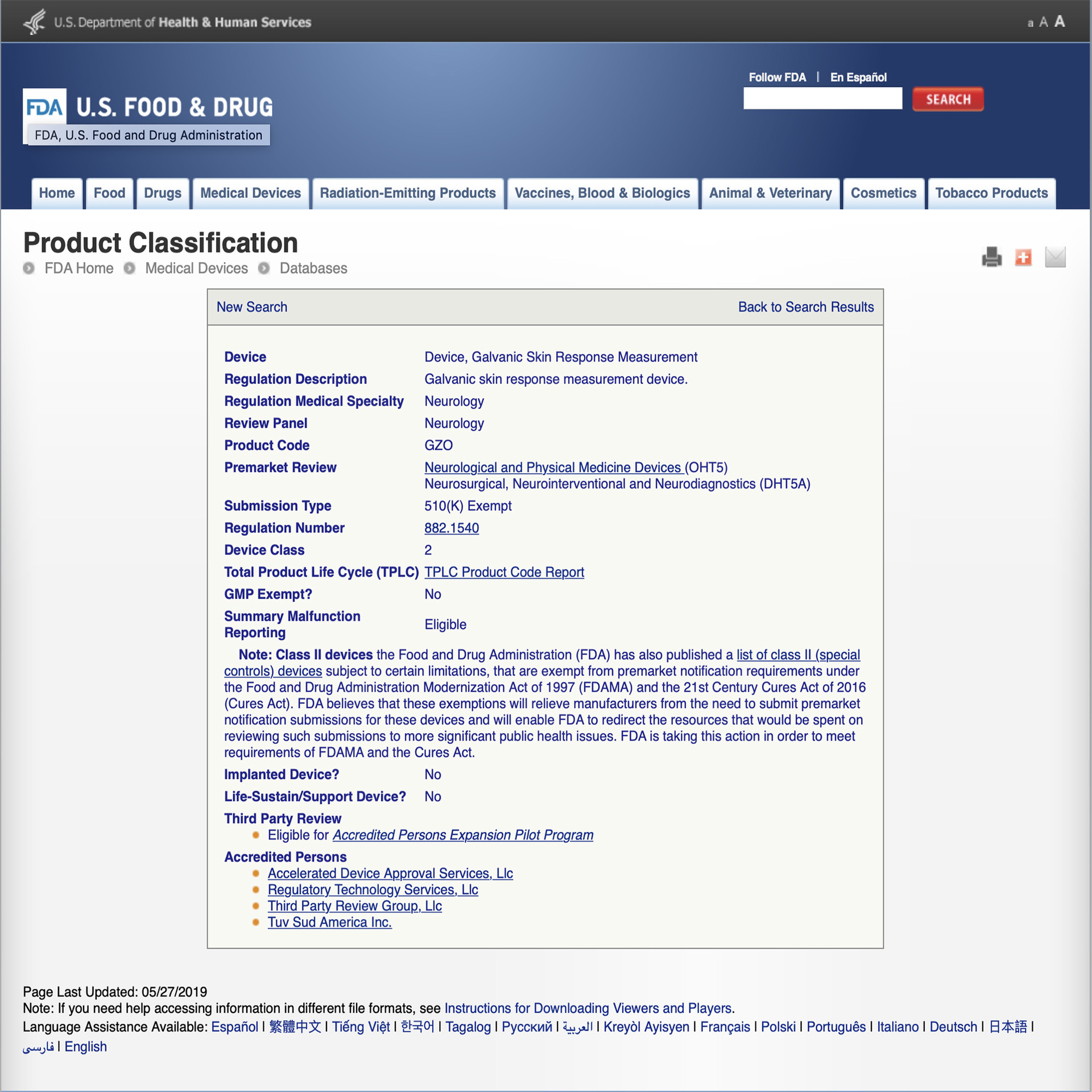
BioMeridian ® is the worlds leading producer and manufacturer of Meridian Stress Assessment Systems (MSAS® ) or EAV class II medical devices. Each MSAS® is manufactured to ISO 9001:2000 standards, the computer boards are manufactured to ISO 9002 standards and utilise programable chips. BioMeridian’s devices are listed with the FDA with the 510(k) number K993824. BioMeridian ® systems that include the "IMAG" for frequency signal output are manufactured to be FCC Compliant.
The MSA Pro is registered with the FDA as a Neurological and Physical Medicine Devices (OHT5)
WANT TO FIND OUT MORE ?
PLEASE LEAVE YOUR DETAILS HERE
LEAVE US A MESSAGE
Just fill out the form below and we will get back to you shortly.
Why Sensitive Foods?
We are experts in helping identify potential Food intolerance and triggers that can contribute to ongoing health issues. But more importantly than just identifying potential triggering events we are experts in helping de-mystify the jigsaw puzzle that helped create these conditions in the first place with detailed personal and family history analysis. We always strive to find out why your problem developed - not just fix the end symptoms.
Our Services
Clinical Nutrition
Food Intolerance Testing and Management
Immune Health Management
Gut Health Management
Planned Recovery Programs
Catering To All Ages From 3 Months Old
Company Information
Fusion 21
ABN :56 970 090 739
Douglas Mizzi - Clinical Nutritionist
ATMS 8070, P.Nutri (Q.I.N.S)
0433 121 736 Direct or Text
1300 55 69 79 General Office
PO Box 133 Mooloolaba
Privacy & Legal Policies HERE
Sensitivefoods.com - Journey To A Better You.